Areas of Focus
Paroxysmal Supraventricular Tachycardia (PSVT)
PSVT is caused by an abnormality in the electrical system of the heart. People with the condition of PSVT have unexpected episodes of rapid heart rate that start and stop suddenly.
A normal resting heart rate is between 60 and 100 beats per minute. During an SVT episode, a patients’ heart rate increases dramatically, sometimes exceeding 250 beats per minute. While experiencing an episode of SVT, symptoms may include palpitations, sweating, chest pressure or pain, shortness of breath, sudden onset of fatigue, fainting, and anxiety.
There are no FDA-approved therapies for the acute termination of PSVT in an at-home setting, and all approved therapies require intervention in a monitored, acute care setting, such as an emergency department.
Frequency and Severity
PSVT can appear at any time in a patient’s lifetime and may occur in otherwise healthy patients. The frequency and severity of episodes vary from patient to patient and can even vary within an individual patient. Our market research shows that patients living with PSVT experience a median of 12 to 15 episodes per year despite up to two-thirds of them taking daily prescription medications to prevent them. Episodes may last anywhere from minutes to hours and can be debilitating for patients, leaving them unable to focus on family or work. Market research also shows that approximately 60% of patients with PSVT experience multiple 10+ minute moderate to severe episodes per year, and approximately 40% experience an average duration of at least 30 minutes per episode.
While PSVT is not typically life threatening, the uncertainty of when an episode will occur and how long it will last can significantly impact patient quality of life. Researchers have noted that up to 27% of patients living with PSVT stopped driving for fear of temporary loss of consciousness. Furthermore, patients often feel anxious in anticipation of future PSVT episodes and frustrated by their lack of a sense of control over their disease.
Prevalence and Incidence
PSVT affects approximately two million Americans and results in as many as 300,000 new diagnoses and between 650,000 and 1,000,000 healthcare claims per year in the US, including emergency department visits, hospital admissions, and ablations. These estimates are based on IQVIA Pharmetrics Plus 2019 Commercial claims for patients <65yo and Medicare LDS 5% for patients >65yo (ICD: I47.1).
A 2021 retrospective, observational, longitudinal analysis published in the Journal of Cardiovascular Electrophysiology utilized claims data from both Medicare and private insurers to estimate the treated prevalence and incidence of PSVT in the U.S. over a nine-year period (2008–2016). This study found that the U.S. treated prevalence for PSVT was 1.3–2.1 million, with approximately 190,000–310,000 new cases each year. Women are affected at a higher rate, comprising two out of every three patients. Except for atrial fibrillation (AFib) and atrial flutter (AFL), the most common comorbidities identified in patients with PSVT were chronic pulmonary disease, diabetes mellitus and heart failure, each of which was more common in individuals aged 65 or older and in those with AFib/AFL.
Compared to past analyses, the updated PSVT prevalence and incidence data show more than twofold increases. Earlier published sources documenting the demographics, clinical characteristics and epidemiology of PSVT rely on a single medical encounter confirmed by an ECG. Thus, these studies estimate the prevalence and incidence of PSVT patients presenting to the healthcare system during an active SVT episode. They likely significantly underestimate PSVT prevalence due to the episodic nature of the disease, the variability in the duration of SVT episodes and the fact that the majority of SVT episodes are experienced outside the health care setting.
This retrospective, observational, longitudinal analysis—which accounted for misdiagnoses and overlap with symptoms of AFib/AFL—generated a more definitive, updated treated prevalence estimate for PSVT within the U.S. population. Given that the minority of adjudicated PSVT was determined to originate from the PSVT code in prior studies, there are likely additional PSVT patients receiving other diagnosis codes that are not included in the 1.3–2.1 million treated prevalence estimate. patients receiving other diagnosis codes that are not included in the 1.3-2.1M treated prevalence estimate.
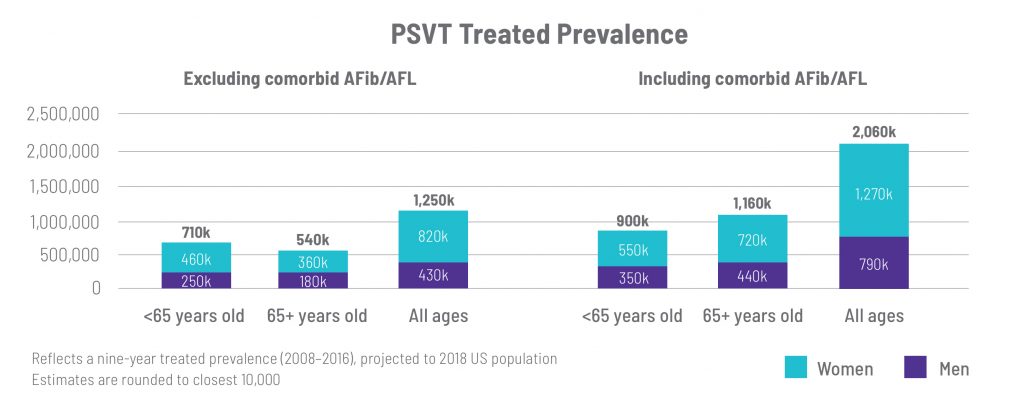
Those living with PSVT may have other comorbid arrhythmias, such as AFib or AFL. Excluding patients with comorbid AF/AFL leads to a conservative estimate of PSVT treated prevalence in the U.S. (~1.3M), while including those with comorbid AF/AFL suggests a U.S. treated prevalence of approximately 2.1M.
Healthcare Economics
Current treatment for PSVT consumes significant healthcare resources. Research published in the American Journal of Cardiology in 2020 shows that costs for patients rose significantly in the pre-diagnosis year due to the difficulty of obtaining an accurate diagnosis. In the year following diagnosis, costs triple for those less than 65 years of age and double for those over 65 years of age, compared to matched controls. Total healthcare expenditures in the year following PSVT diagnosis ranged from $20,000-$30,000 per patient, significantly higher than the expenditures observed for patients without PSVT (~$6,500 per patient). Significant increases for both age groups were noted for emergency department visits. For those less than 65, the average cost of hospitalizations doubled as their inpatient rates quadrupled. Of note, catheter ablations following diagnosis represent only 23% of this increased spend, meaning the majority of costs are unrelated to ablations.
Based on market data on file, we estimate approximately 100,000 catheter ablations and 140,000 to 525,000 Emergency Department visits for PSVT occur each year and 40,000 to 120,000 inpatient hospitalizations, driving the majority of the approximately at least $5 billion spent annually in the U.S. on the management of PSVT.
Looking to the broader landscape of economic burden on our health system from cardiac treatments, a 2021 study released in the American Heart Journal found patients <65 living with PSVT can cost the health care system a comparably similar amount to patients with AFib—the most common arrhythmia diagnosed in U.S. clinical practices. This study followed patients for up to six years post-diagnosis. Similar to the American Journal of Cardiology study, this data showed that costs never return to baseline, which indicates a need for more treatment options in long-term PSVT management.
How costly is PSVT to our healthcare system?
PSVT costs rise significantly from the year before to after diagnosis in both younger (<65 years) and older (≥65 years) patients
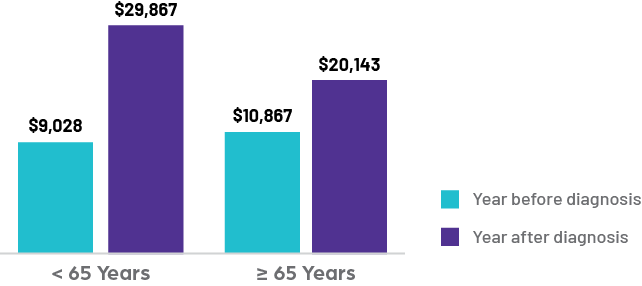
Cost Increases Include:

<65 years: 4x increase
≥65 years: 2x increase

<65 years: 2.5x increase
≥65 years: 33% increase

<65 years: 13% of patients
≥65 years: 3% of patients
Gender Disparity
Symptoms experienced by PSVT patients are commonly misdiagnosed as anxiety or panic attacks, especially in women. Compared to men, it can take women more outpatient and emergency department visits to be correctly diagnosed. Following diagnosis, women are more likely to be treated with medical therapy to manage symptoms, while men are more often referred for an ablation.
Standard of Care
The current standard of care for the acute treatment of PSVT is an intravenous (IV) injection of adenosine, usually given in a hospital or emergency department. Adenosine blocks conduction over the atrioventricular (AV) node, a piece of electrical tissue that regulates the rhythm in the heart, interrupting the arrhythmia and restoring the heart rate to normal. Some patients report experiencing chest tightness, flushing, and even a sense of impending death with adenosine. Adenosine is eliminated from the body in less than one minute and cannot be self-administered by the patient, as it requires IV access.

Prior to the approval of adenosine, PSVT was treated with IV calcium channel blockers, such as verapamil or diltiazem, that also slow conduction over the AV node within minutes. These drugs bear the risk of excessive slowing of the heart rate and lowering of blood pressure. In-hospital IV administrations are associated with higher health care costs and are inconvenient for the patient.

Many patients take daily oral medications, such as beta blockers, calcium channel blockers, or antiarrhythmic drugs, in an attempt to prevent or reduce the frequency and duration of future PSVT episodes. This can still result in “breakthrough” episodes requiring visits to the emergency department. Some patients discontinue oral medication due to side effects. For instance, taking beta blockers chronically may cause sexual dysfunction or fatigue, and long-term use of verapamil may cause constipation.
Patients may also try taking one of these medications when they experience an episode. This is referred to as a “pill-in-the-pocket” approach, which will not prevent episodes from occurring, but may help shorten their duration. Market research suggests that approximately 25-30% of PSVT patients attempt the pill-in-the-pocket approach to manage their episodes.

The only potentially curative treatment for PSVT is ablation, an invasive, interventional procedure, which cauterizes the short circuit that causes the abnormal rhythm. This procedure burns or freezes the heart’s abnormal electrical tissue with catheters that are run through the patient’s groin vessels and into the heart. Approximately 10-15% of patients living with PSVT ultimately receive a catheter ablation.
Let’s Work Together
We collaborate with leading researchers, academic institutions, scientific societies, and patient support organizations who are like-minded in the pursuit of PSVT knowledge and education. If you are interested in collaborating with us, please email us at info@milestonepharma.com.
- Brugada, J., European Heart Journal. 2019;41(5):655-720.
- Chew, D. S., Am Heart J. 2021;233:132-140.
- Colucci, R. A., Am Fam Physician. 2010;82(8):942-952.
- Go, A. S., J Am Heart Assoc. 2018;7(14):1-7.
- Lessmeier, T. J., Arch Intern Med. 1997;157(5):537-543.
- Milestone Pharmaceuticals data on file: internal estimates based on market research and longitudinal analysis of Truven/Marketscan and Medicare claims data.
- Orejarena, L. A., J Am Coll Cardiol. 1998;31(1):150-7.
- Page, R., J Am Heart Assoc. 2016;133(14):e506-e574.
- Rehorn, M., J Cardiovasc Electrophysiol. 2021;32 (in press).
- Sacks, N. C., Amer J of Card. 2020;125(2):214-221.
